Clinical Evaluation requirements have increased dramatically since the release of MEDDEV 2.7.1 Rev 4 in 2016 and the MDR 2017/745 in May of 2017. The process now involves two documents; the Clinical Evaluation Plan (CEP) and Clinical Evaluation Report (CER). Some Notified Bodies were requiring Clinical Evaluation documents to be compliant to MEDDEV 2.7.1 Rev 4 as early as January 2017. However, the MEDDEV is a guidance document that is not legally binding. Therefore, some Notified Bodies have extended the deadline for implementation of the MEDDEV to match that of the MDR. That said, Notified Bodies will still expect to see a plan on how the new regulations will be implemented. Full compliance to both the MEDDEV and MDR is expected by May 26, 2020. As such, the clinical team at Qserve has been busy providing trainings at manufacturer locations and writing MEDDEV and MDR compliant CEPs and CERs. On February 27th, Jasmin Hunter presented a 1-hour version of a 2.5-day Clinical Evaluation Workshop for ASQ Biomedical Division of the New England Discussion Group. One hour was not enough time to provide the hands-on experience writing portions of a CER, but some tips and tricks on the essential elements were provided. This blog will cover a few of those elements in more detail.
Sufficient Clinical Data Versus Sufficient Clinical Evidence
Many of the CERs that Qserve evaluates have plenty of data. What they often lack is an analysis of the data presented. Data on its own is meaningless without context. Data only becomes evidence when a specific viewpoint is added. The process of clinical evaluation starts with the origin of a research question based on the data at hand. The final step is to evaluate that data to determine if you have sufficient clinical evidence to support conformity to GSPRs 1 and 8 (at a minimum). Evidence is built by telling the story about the data. For example, if adverse event data from PMS and clinical literature are used together to determine the occurrence rate of clinical risks associated with the device, there is a higher degree of confidence that the information is correct. The two sources analysed together create evidence. If the two sources disagree, more data must be collected. This process is done for each performance and safety requirement, as well as the claims and benefits outlined in the CEP. Data from bench-testing, PMS, clinical investigation, and clinical literature can be analysed together to build sufficient clinical evidence.
How do you get started?
The process of clinical evaluation requires input from many different departments within an organization. The CEP should provide a detailed story as to why the device was designed and what it is designed to do. The MEDDEV Rev 4 provides a great description of how to define the scope of the clinical evaluation based on the Essential Requirements (now General Safety and Performance Requirements under the MDR) that need to be addressed from a clinical perspective and the nature and history of the device in Section 7. The purpose of the plan is to assess the data already available to support the intended use, performance, safety, benefits and claims of the device.
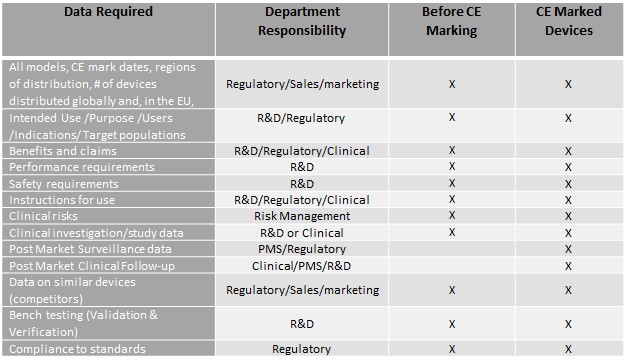
If you have been assigned to write a clinical evaluation plan and report, your first step is to contact all the relevant departments in your organization to collect the required information for the device. The structure of every manufacturer is different, but your organization will have similar departments to those listed in the table below. The data you will need for a new device is slightly different than what is needed for an already CE marked device in that after CE marking, Post Market Surveillance (PMS) data and Post Market Clinical Follow-up data must be included in the CER.
After assessing data available, research questions are created to specifically address gaps in data or to support existing data. Data can then be pulled from clinical literature, device registries, healthcare databases, or disease specific databases to answer the defined research questions. Clinical data from literature is a required first step. The other options listed can be used to enhance the data found or to fill in gaps if data is not available in the literature.
Establishing State-of-the-Art (SToA)
A critical component of the clinical evaluation process is establishing the state-of-the-art in medicine (clinical SToA) for the disease that the device is intended to treat, monitor, or diagnose. The discussion of SToA should start in the CEP with the background of the disease associated with the device, including all of its forms and severities, as well as, any alternative treatment options, including medicinal treatments. Applicable standards and guidance documents related to the device being evaluated should also be included to provide the technical aspect of SToA.
This discussion provides a framework by which to verify that clinical risks associated with the device being evaluated are comparable to other treatments for the disease in similar patient populations. To that end, the SToA in the CEP should also include acceptance criteria for data on performance, safety, benefits, and claims, as well as, the benefit-risk ratio and acceptability of side-effects evaluated in the clinical evaluation. This will serve as a justification for the amount of clinical evidence necessary to demonstrate conformity with the relevant general safety and performance requirements as required by MDR Article 61, point 1.
The SToA in the CER will build on this discussion to include prognosis if the disease is left untreated, and the risks and benefits of the alternate devices discussed in the CEP. Other information required for SToA in the CER includes:
- Frequency of device use in the general population;
- Diverging opinions of professionals concerning device use and treatment options;
- Unmet needs of device technology;
- Clinical hazards (risks) to at risk patients in the general population – pediatric, elderly and pregnant patients.
There are many more aspects of the clinical evaluation process that cannot be captured in a blog. Please contact us if you are interested in a training session at your organization.
Profile: